Firmagon® INJECTION
FIRMAGON® (degarelix for injection) IS ADMINISTERED AS A DEEP SUBCUTANEOUS INJECTION IN THE ABDOMINAL REGION EVERY 28 DAYS1
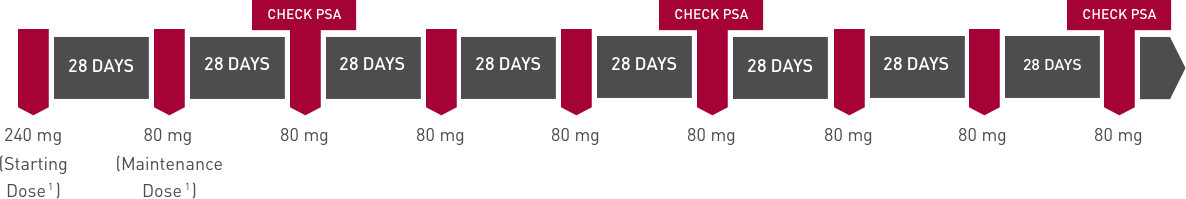
- To determine the lowest PSA achieved, multiple measurements of PSA will be required
Starting Dose
240 mg: 2 x 120 mg
(2 x 3 mL injection)
Maintenance Dose
80 mg: 1 x 80 mg
(1 x 4 mL injection)
Use product-specific HCPCS code J9155 for both doses of FIRMAGON
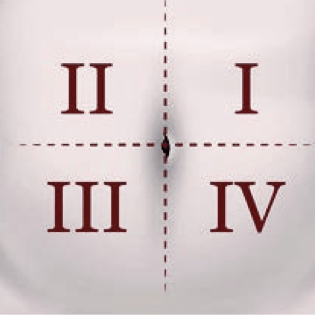
The injection site should vary periodically
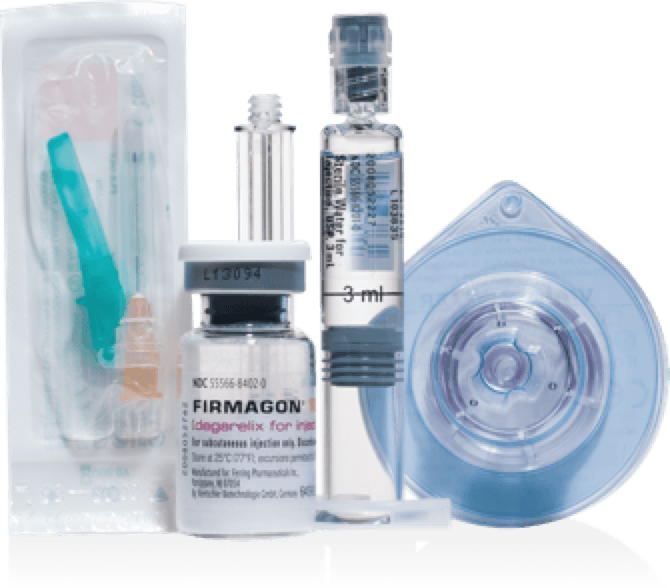
THE FIRMAGON® PREFILLED KIT
DESIGNED FOR EFFICIENT RECONSTITUTION
Requires reconstitution but not refrigeration.
PREPARING THE FIRMAGON® DOSE1
FOLLOW ASEPTIC TECHNIQUE
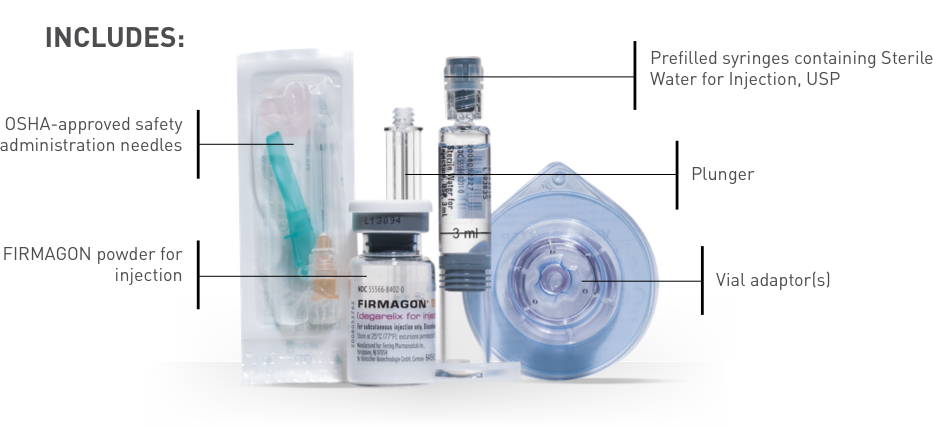
Use only the materials provided in kit for reconstitution and injection.
Caution should be exercised in handling and preparing the solution of FIRMAGON. To minimize the risk of dermal exposure, always wear impervious gloves when handling. If solution contacts the skin, immediately wash the skin thoroughly with soap and water. If FIRMAGON contacts mucous membranes, the membranes should be flushed immediately and thoroughly with water.
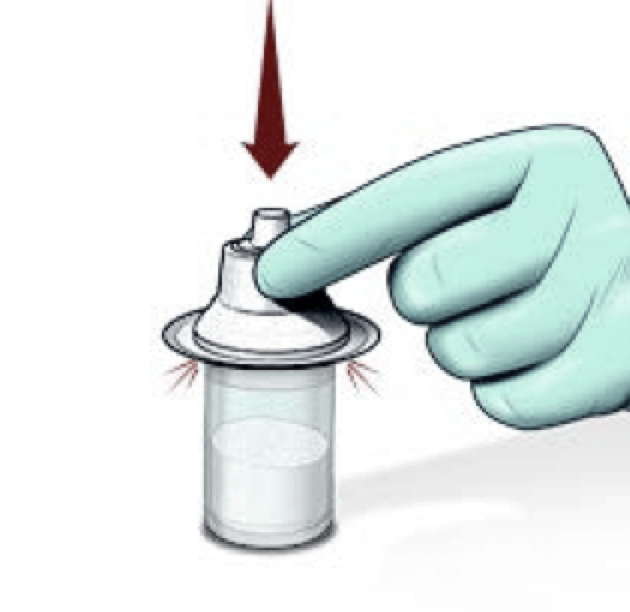
Attach the vial adaptor
IMPORTANT: Do not touch the top of the vial after wiping. Do not use if there is no powder in the vial or the Sterile Water for Injection, USP is discolored.
Assemble the syringe
NOTE: You will only feel light resistance when screwing the plunger rod into position.
IMPORTANT: Do not pull off the Luer lock adaptor.
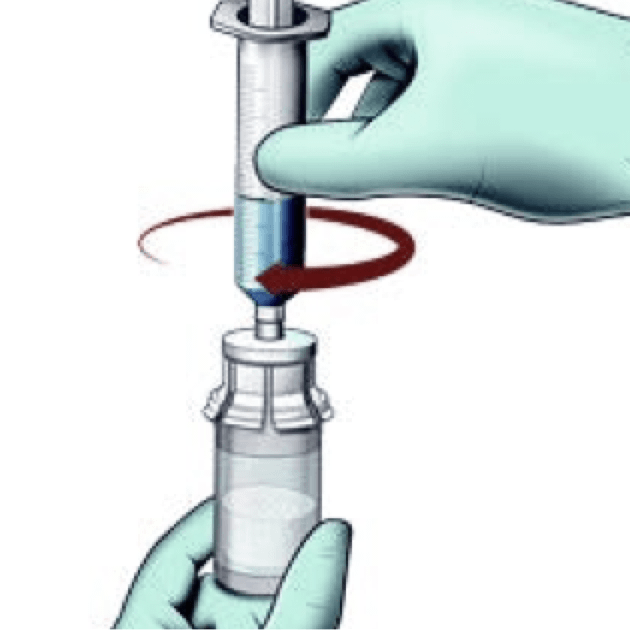
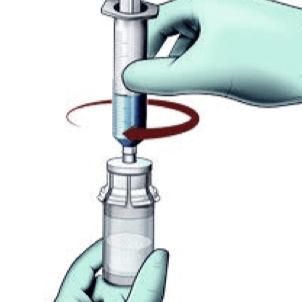
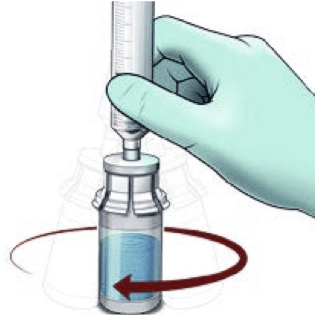
Reconstitute
- Do not shake the vial as this will cause bubbles
- Reconstitute just prior to administration
- If the powder adheres to the side of the vial, tilt the vial slightly. A ring of small air bubbles on the surface of the liquid is acceptable
- Reconstitution time can take up to 15 minutes but usually takes a few minutes
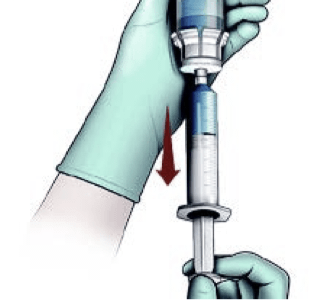
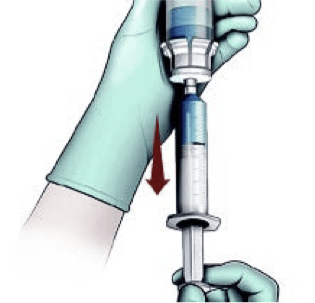
Transfer liquid to syringe and prepare for injection
NOTE: Reconstitute just prior to administration.
ADMINISTERING THE FIRMAGON® DOSE1
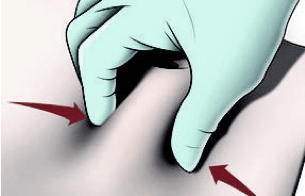
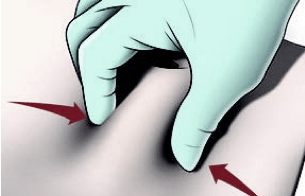
Prepare the patient
- Do not inject in areas where the patient will be exposed to pressure, such as the areas around the belt or waistband or close to the ribs
- Vary the injection site periodically during treatment to minimize patient discomfort
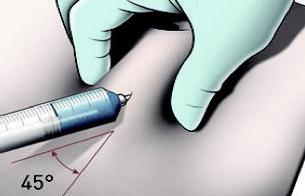
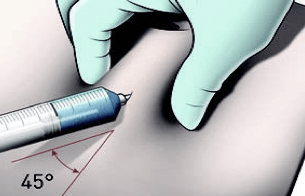
Perform the injection
IMPORTANT: If blood appears in the syringe, the product should not be injected. Discontinue the injection and discard the syringe and the needle (reconstitute a new dose for the patient).
IMPORTANT: Trauma can occur in the dermal layer if the medication is injected too fast.
IMPORTANT: Once the needle is removed, continue to hold the skin for ≈15 seconds to allow the injection tract to close. Then, release the skin.
WHEN THE INJECTION IS COMPLETE, PLACE THE NEEDLE GUARD OVER THE NEEDLE UNTIL IT CLICKS, AND DISPOSE INTO A SHARPS CONTAINER.
Post-injection care instructions for patients
Here is a quick rundown of what to tell patients right after their injection:
- Do not rub or scratch the injection site
-
- Keep the site clean and wear loose clothing to avoid irritating it
- There can be a lump at the injection site and redness, soreness, and discomfort for a few days
- The lump is a depot—or medication deposit—that supplies the body with a continuous release of FIRMAGON over time
-
- The lump will eventually subside
- Schedule the next dose of FIRMAGON before your patient leaves the office
Abbreviations: HCPCS = Health Care Procedure Coding System; OSHA = Occupational Safety and Health Administration; USP = United States Pharmacopeia.
REFERENCE: 1. FIRMAGON® [package insert]. Parsippany, NJ: Ferring Pharmaceuticals Inc.